The nitrogen Cycle
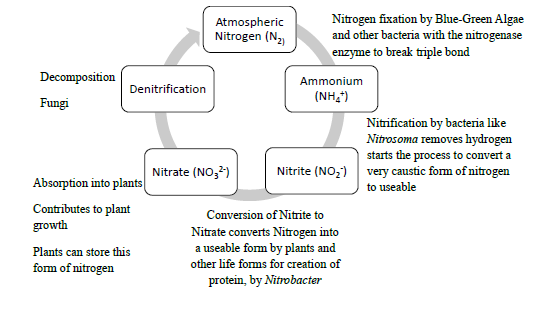
Nitrogen is an essential element to life on earth. It naturally occurs in our atmosphere. Our air on earth is approximately 78% nitrogen, 20% oxygen, 1% argon, 0.04% carbon dioxide, and small amounts of other gases. Nitrogen makes up parts of DNA, so is useful to organisms. Bacteria convert nitrogen into useable forms.
Naturally occurring Stable Isotopes of Nitrogen
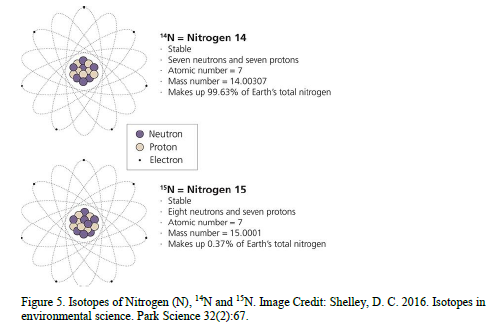
In addition to these naturally occurring compounds, as an element, Nitrogen (N) has two stable isotopes (Figure 5). Stable isotopes have a nucleus that is stable indefinitely and they do not emit radiation (Shelley 2016). Different isotopes of the same element all take part in chemical reactions, but at slightly different rates. “Lighter” isotopes move around more easily than “heavier” ones. This differential movement is called fractionation, and it affects the ratio of isotopes present in any given sample of water, soil, rock, tissue, or air (Shelley 2016).
Stable isotopic data can provide evidence of dominant nitrogen sources incorporated in water samples or in plant tissues. The fractionation processes of N (into δ14N and δ15N) are very important to consider for accuracy of the source identification (Xue et al. 2009). The stable isotope method relies on the assumption that bacteria preferentially take up δ14N first, and δ15N after.
Stable isotopic data can provide evidence of dominant nitrogen sources incorporated in water samples or in plant tissues. The fractionation processes of N (into δ14N and δ15N) are very important to consider for accuracy of the source identification (Xue et al. 2009). The stable isotope method relies on the assumption that bacteria preferentially take up δ14N first, and δ15N after.
Stable nitrogen isotope analysis
It is based on the principle that nitrogen has two stable isotopes that exist, δ14N, which is typically found in natural sources, and δ15N, which is typically found in artificial, or anthropogenic sources, such as sewage.
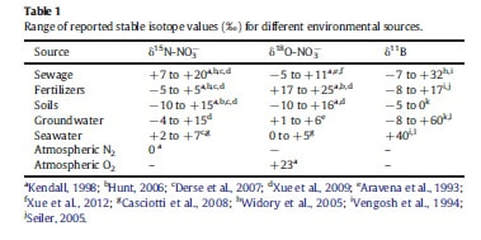
The ratio of δ14N to δ15N, can suggest if the nitrogen in the environment is from sewage (including cesspool, septic tank) or from agriculture (fertilizers, herbicides, pesticides) and can be seen in the Table 1 below (Wiegner et al. 2016).
Read an article about similar research here: Puako Study
The ratio of δ14N to δ15N, can suggest if the nitrogen in the environment is from sewage (including cesspool, septic tank) or from agriculture (fertilizers, herbicides, pesticides) and can be seen in the Table 1 below (Wiegner et al. 2016).
Read an article about similar research here: Puako Study